recomLine CMV
against Cytomegalovirus (CMV) and for the determination of CMV-IgG avidity in human serum or plasma.
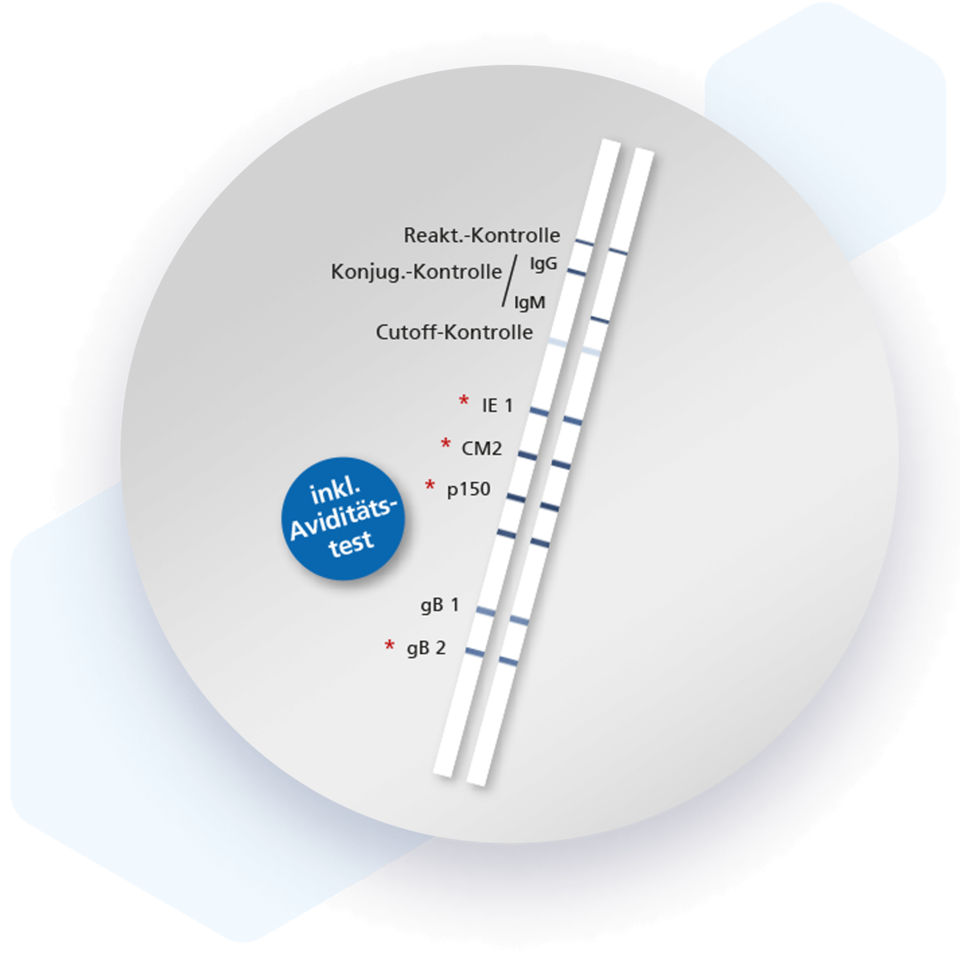
Mikrogen recomLine CMV tests are serological, qualitative in vitro line immunoassays based on recombinantly produced antigens. The test is used as a confirmatory test of positive and inconclusive screening results. The low predictive value of a positive CMV IgM detection, due to persistent IgM or recurrent infection, makes reliable diagnosis of CMV infection particularly difficult. Using the recomLine CMV, IgG, IgM as well as the avidity of IgG antibodies can be determined. Phase-specific antigens and avidity antigens (patented by MIKROGEN on the Immuno-Line Assay) allow detection of antibodies in all phases of infection. In addition to viral load, the recomLine CMV can be used in pregnancy diagnostics as well as in preoperative diagnostics in transplant patients to detect CMV infection and to determine the time of infection.
The recomLine CMV tests provide a clear answer to the question of what effects the present infection may have on the health status of the individual patient and thus support an optimal therapy decision.
Products
Advantages
- Highly reliable patient management through the use of phase-specific antigens that characterize the time of infection
- Reliable detection of pregnancy-related CMV infections
- Simple and clear interpretation due to easy-to-read banding
- Partial and full automation, software-based evaluation (recomScan) and integration with to laboratory information system possible
- Highest sensitivity and specificity through the use of recombinant antigens:
| Antigen | Reading frame/protein | Natural function | Size [kDA] |
| IE 1 | UL 123 / IE 1/1 | Non-structural protein, immediate early protein | 53 |
| CM2 | UL44, UL57 / p52 (DBP) | Non-structural protein | 45 |
| p150 | UL32 / pp150 | Tegument protein | 50 |
| p65 | UL 83 / pp65 | Tegument protein | 31 |
| gB 1 | UL 55 / gB | Membrane glycoprotein gB | 25 |
| gB 2 | UL 55 / gB | Membrane glycoprotein gB | 18 |
Safe and reliable
Validity check by integrated, strip-specific cutoff and antibody class controls
Flexible and compatible
Combination of all Microgen line immunoassays possible - uniform processing and interchangeable reagents
High Standard
CE mark: Our recomLine CMV tests meet the high standard of the EC Directive 98/79/EC for in vitro diagnostic medical devices.