recomLine EBV
Line immunoassays for the detection of IgG, IgA and IgM antibodies
against Epstein-Barr virus (EBV) in human serum and plasma
against Epstein-Barr virus (EBV) in human serum and plasma
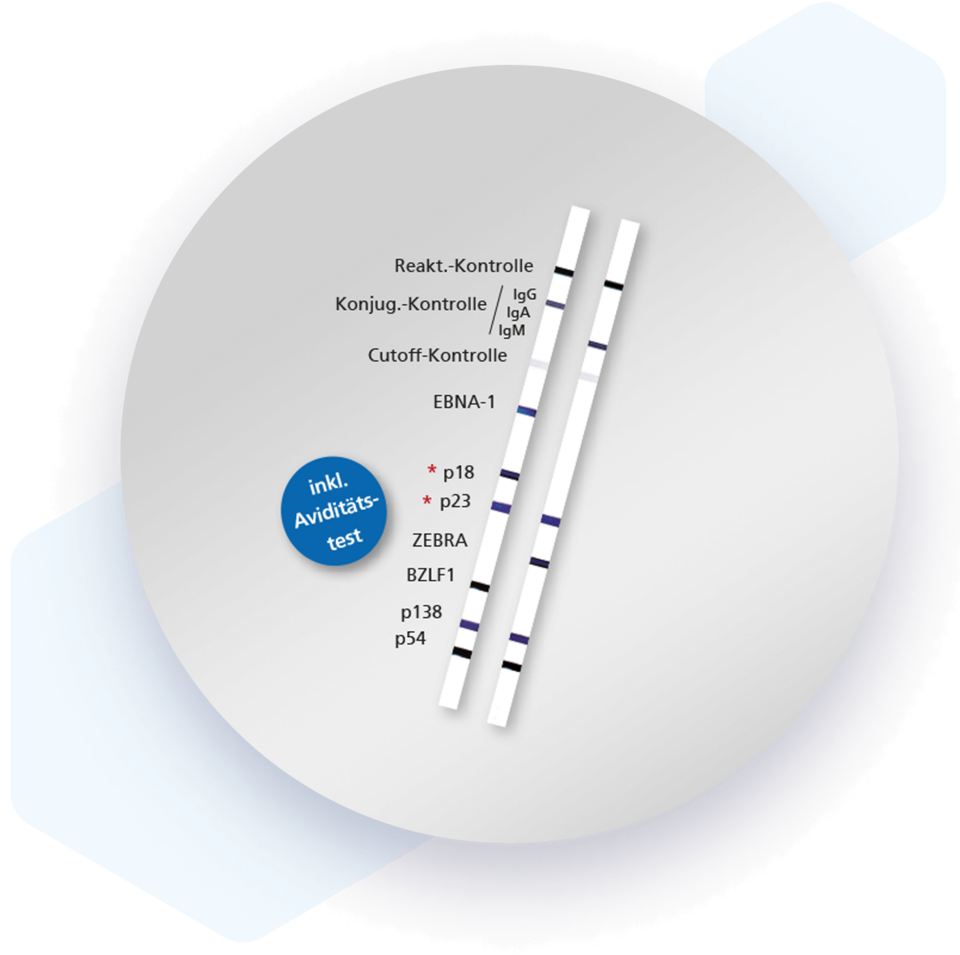
Mikrogen recomLine EBV tests are serological, qualitative in vitro line immunoassays based on recombinantly produced, highly specific and characteristic EBV antigens. They are designed as screening tests. The line assay technique allows the detection and identification of IgG and IgM antibodies against the different antigen classes in one approach and at one glance.
Products
Advantages
- No disturbing interference with anticellular antibodies
- Patented p18Mikrogen as additional IgG marker for past infections
- New antigens: Highest sensitivity by BZLF1 in IgG and ZEBRA in IgM detection, in the early phase of acute infections
- In more than 95% of cases, past EBV infections can be correctly identified with the recomLine EBV IgG strip alone
- Simple and meaningful avidity determination - patented
- Separate detection of IgG and IgM antibodies
- Simple and clear interpretation due to easy-to-read banding
- Partial and full automation, software-based evaluation (recomScan) and integration with laboratory information system possible
- Highest sensitivity and specificity through the use of recombinant antigens:
| EBV antigen groups | Abbreviation | Recombinant antigen | Size of recombinant antigen |
| Nuclear antigen | EBNA-1 | p72 | 45 kDaL |
| Virus capsid/structural antigen | VCA | p23 p18 | 23 kDal 18 kDal |
| "Immediate Early Antigen" | IEA | ZEBRA (Peptid) BZLF1 | 1,6 kDal 30 kDal |
| "Early Antigen" | EA | p54 p138 | 54 kDal 40 kDal |
Safe and reliable
Validity check by integrated, strip-specific cutoff and antibody class controls
Flexible and compatible
Combination of all microgen line immunoassays possible - uniform processing and interchangeable reagents
High Standard
CE mark: Our recomLine EBV tests meet the high standard of the EC Directive 98/79/EC for in vitro diagnostic medical devices.