recomLine Yersinia
against Y. enterocolitica and Y. pseudotuberculosis in human serum or plasma
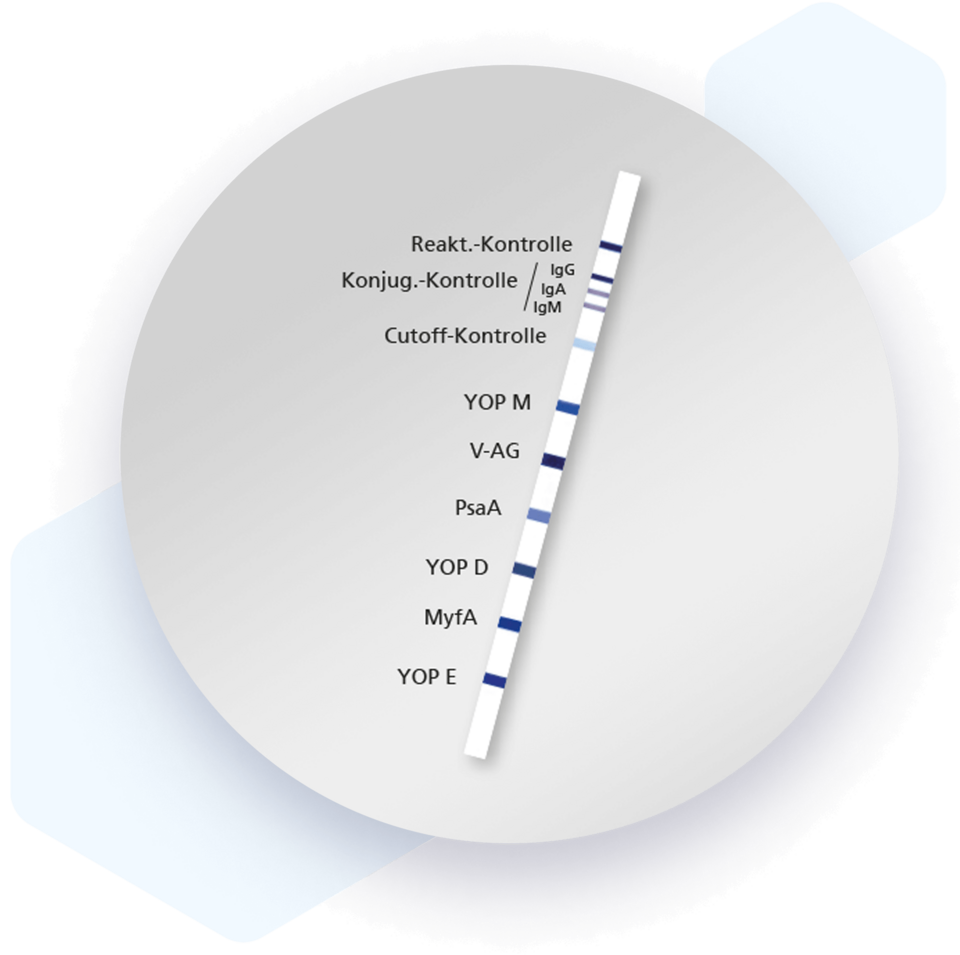
For the Mikrogen recomLine Yersinia assays, the plasmid-encoded virulence proteins (Yersinia Outer Proteins) localized at the cell surface and adhesins for serological species differentiation between Y. enterocolitica and Y. pseudotuberculosis are produced recombinantly. These proteins are expressed only by human pathogenic Yersinia strains.
The recomLine Yersinia tests allow the detection of long-standing Yersinia infections and are thus ideally suited to identify Yersinia-induced immunopathological complications and chronic yersinioses. The detection of IgG and IgA antibodies can be an important aid in suspected Yersinia-induced arthritis.
Products
Advantages
- Detection of all human pathogenic Yersinia by means of Yersinia Outer Proteins (YOPs)
- Differentiation between Y. enterocolitica and Y. pseudotuberculosis infection serologically possible by using new species-specific Yersinia antigens (PsaA, MyfA)
- No cross-reactions to Brucella and other pathogens, and no interference by LPS
- Separate detection of IgG, IgA and IgM antibodies - antibody class control on the test strip - helpful in case of suspected Yersinia-induced arthritis
- Partial and full automation, software-based evaluation (recomScan) and connection to laboratory information system possible
- Highest sensitivity and specificity by using different, recombinant Yersinia antigens:
| Antigen | Description |
| YOP M | Yersinia outer protein |
| V-AG | Yersinia virulence factor |
| PsaA | Adhesin (specific for Y. pseudotuberculosis) |
| YOP D | Yersinia outer protein |
| MyfA | Adhesin (specific for Y. enterocolitica) |
| YOP E | Yersinia outer protein |
Safe and reliable
Validity check by integrated, strip-specific cutoff and antibody class controls
Flexible and compatible
Combination of all Mikrogen line immunoassays possible - uniform processing and interchangeable reagents
High Standard
CE mark: The recomLine Yersinia test meets the high standard of the EC Directive 98/79/EC for in vitro diagnostic medical devices.